Nordea betegner Moderna’s corona-vaccine som imponerende, fordi dens effektivitet er på 94,5 pct., og fordi den kan opbevares til en lavere temperatur end vaccinen fra BioNTech/Pfizer – henholdsvis køleskabstemperatur og minus 70 grader. Smittetallene udvikler sig vidt forskelligt i Europa.
Daily Corona update: Moderna’s prelimenary phase-3 analysis vaccine results impress
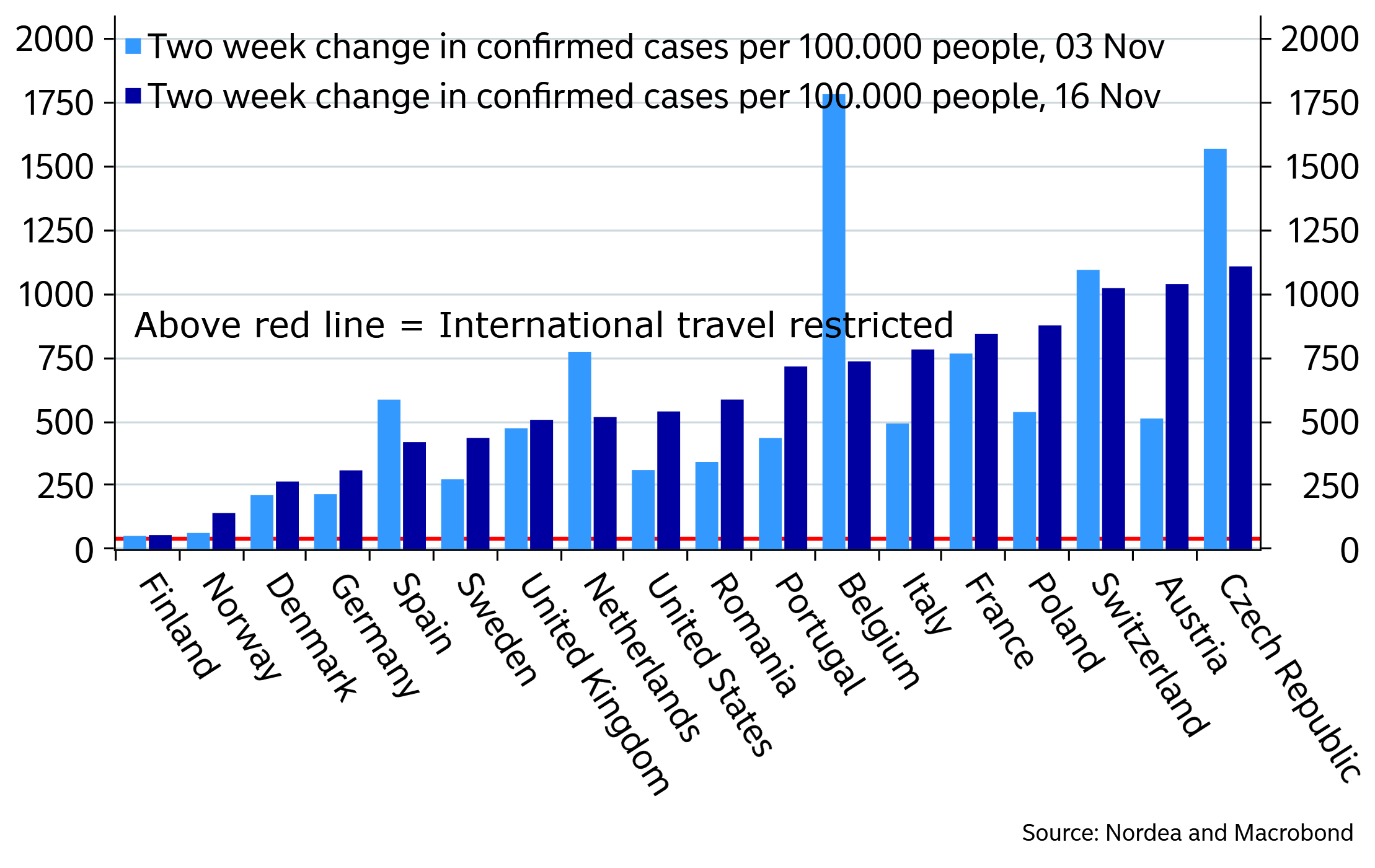
Moderna’s vaccine bid has shown to be 94,5% effective in early analysis of phase-3 trial studies. The US and Austria keeps climbing and Austria now has second most rolling new cases in our analysis.
On the news
Moderna has in an early analysis of their phase-3 trial studies found their bid for a vaccine to be an impressive 94.5% effective.
While not only more effective (keep in mind: early analysis of their phase-3 results) than the Pfizer/BioNTech, it also has other significant properties.
When the Pfizer news broke, it was quickly also remarked that the storage of the vaccine requires ultra-cold (-70C) storage, making widespread distribution more expensive and cumbersome.
The Moderna entry can be stored 30 days in fridge temperature and much longer in freezing temperatures.